505(b)(1)
LXP103
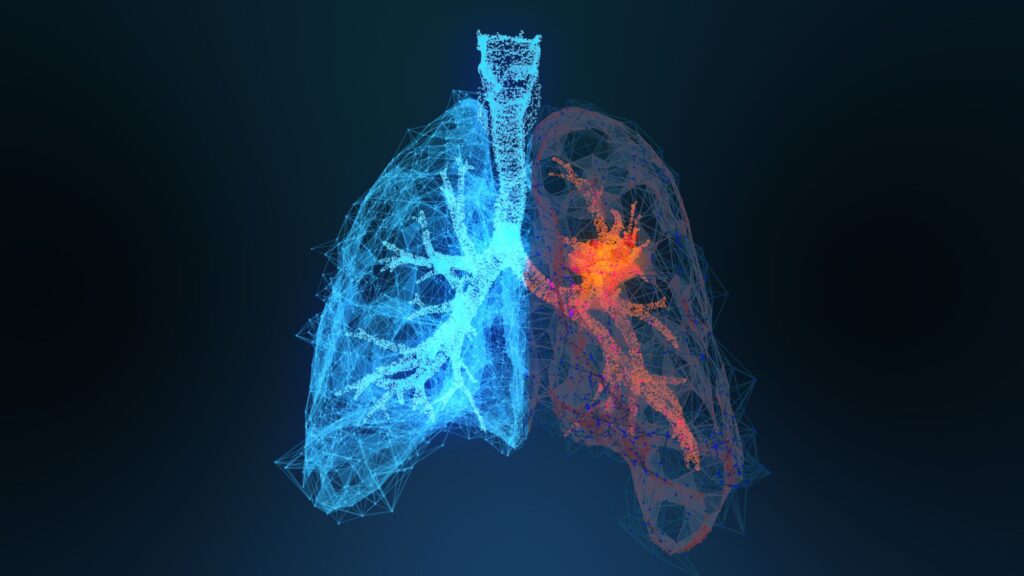
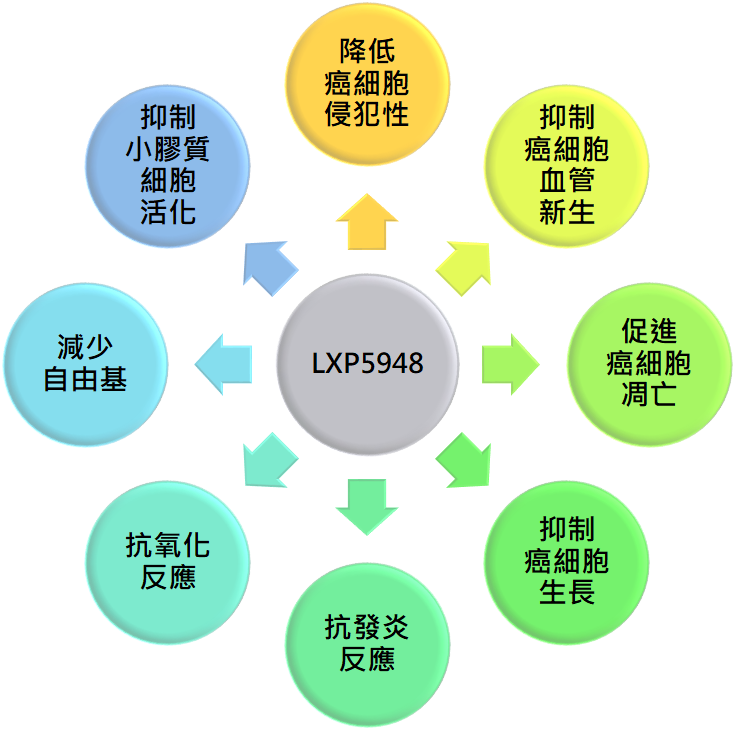
LXP103 has obtained patents in seven countries. Animal studies suggest that LXPA103, when combined with Osimertinib for non-small cell lung cancer, delays resistance to the treatment. Preclinical studies and new formulations are currently underway.
適應症
Non-Small Cell Lung Cancer & Alzheimer's disease
開發價值
Animal test results suggest that LXP103 is more effective when used in combination with Osimertinib, the current third-generation treatment for EGFR-mutated non-small cell lung cancer.
臨床試驗
最新進度
臨床前(Preclinical)
申請(IND)
第一階段(Phase 1)
第二階段(Phase 2)
第三階段(Phase 3)
市場(Market)
臨床前(Preclinical)
專利
已取得台美之專利,其他國家專利申請中
- Taiwan
- United States
