505(b)(1)
LXP103
順應全球趨勢,LXP103為創新化學實體藥物精準調節代謝失調。優勢在於改善代謝功能的同時,兼顧生理機能與長期穩定性。不同於傳統路徑,此藥物提供更溫和、精準的全新選擇,體現了從單一治療邁向全面健康管理的領先研發思維,具備高度差異化潛力。
治療領域:
代謝失調

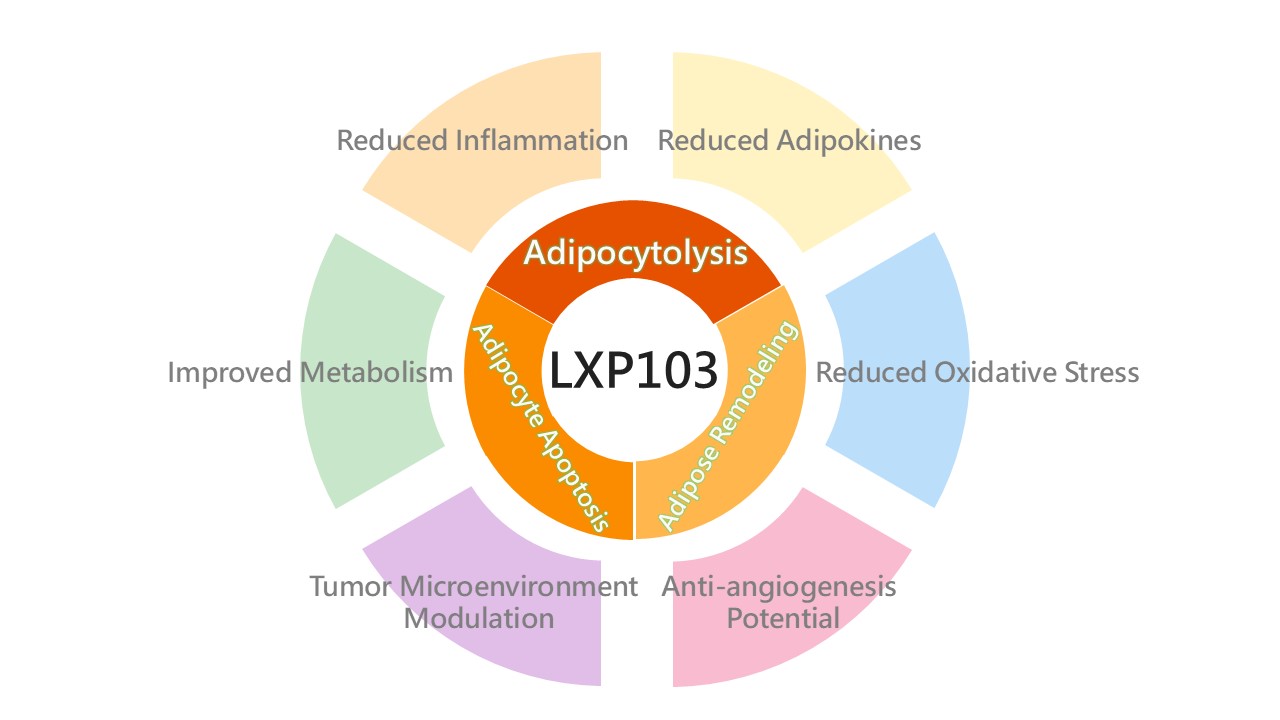
開發價值
隨著全球代謝健康意識提升,市場焦點正從單純的體重下降,演進至追求「生理機能維持」與「長期代謝穩定」的深層管理。臨床亟需能兼顧代謝調節與健康體態的溫和路徑,以填補長期健康管理趨勢下的關鍵缺口。本項創新分子具備全身性調控潛力,研發重點在於透過獨特的代謝調節機制支持生理機能平衡,期望能為代謝失調族群提供更具穩定性與安全性的長期管理方案。
最新進度
開發階段
臨床前
IND 申請
臨床一期
臨床二期
臨床三期
新藥查驗登記
臨床前
專利
已在台灣和美國獲得專利,在其他國家提交的申請仍在進行中。
- 台灣
- 美國
