505(b)(2)
LXP105
The LXP105 serves as a potent "combination backbone" for refractory breast cancer. Utilizing its small molecule’s superior tissue penetration, it synergizes with standard-of-care and immunotherapy to disrupt tumor signaling. This approach enhances therapeutic depth and mitigates resistance risks, establishing a more efficient and precise pathway for clinical combination regimens.
Therapeutic Area:
Triple-Negative Breast Cancer
Current Treatment Methods:
Chemotherapy ➡️ Surgery
Challenges with Current Treatment Methods:
- Triple-negative breast cancer represents approximately 15-20% of breast cancer cases, with only 15% eligible for targeted therapies.
- The objective response rate does not reach 100% (typically 60-90%).
- Tumor shrinkage rate is relatively low (generally around 40-50%).
Development Value
Designed for aggressive tumors, this small-molecule therapy leverages strong tissue penetration to combine with standard or immunotherapies, enhancing efficacy and reducing resistance through synergistic mechanisms.
Latest Progress
Development Stage
Preclinical
IND
Phase I
Phase II
Phase III
NDA
Phase II (Proof of Concept)
Results
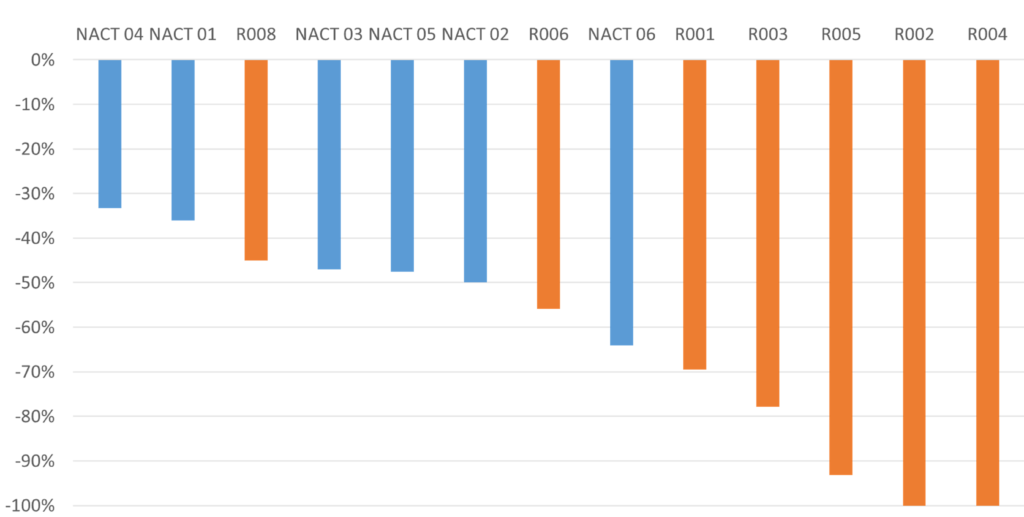
Better tumor reduction, Higher rate of pathological complete response
The LXP105+NACT group showed better tumor reduction, and a higher rate of pathological complete response compared to the NACT group alone.
Significant reduction observed in CT images
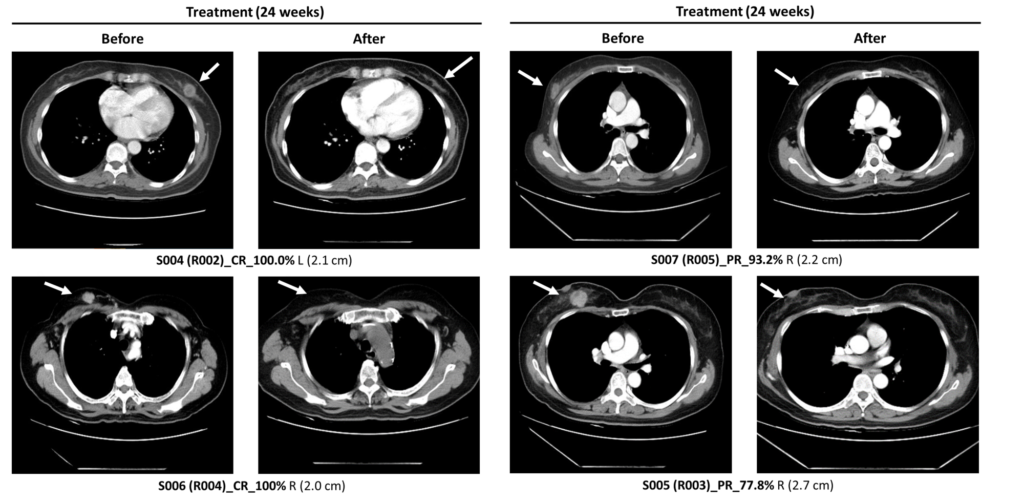
LXP105 combination therapy with NACT better than NACT only
Patents
Patents for cancer-related indications have been granted in Taiwan, the United States, Japan, Australia and European.
- Taiwan
- United States
- Japan
- Australia
- Europe
